Stopping Antidepressants Safely: What Is Deprescribing and How Is It Done?
By Dr. Anindo Mitra | MBBS, MD Psychiatry (JIPMER) | Consultant Psychiatrist, Athena Behavioural Health, Gurugram
Published on dranindomitra.com | Reading time: ~12 minutes
TL;DR — Key Takeaways
• Deprescribing is the planned, supervised reduction or stopping of a medication whose risks now outweigh its benefits. It is part of good prescribing, not the opposite of it.
• The IPS multicentric study (Grover et al. 2014) found that 70% of patients with affective and anxiety disorders in India were co-prescribed a benzodiazepine, making deprescribing a routine clinical competency we need, not an optional one.
• Venlafaxine and paroxetine carry the highest risk of discontinuation symptoms. Long-term use compounds this risk. Most failed attempts to stop fail because the taper was too fast.
• A 2025 Lancet Psychiatry meta-analysis found that slow tapering combined with psychological support was as effective as continuing the antidepressant in preventing relapse in remitted depression.
• Hyperbolic tapering, where the rate of reduction slows as doses get lower, is now the recommended approach for patients with difficulty stopping. The principle: stop slow as you go low.
• Deprescribing is a clinical decision requiring individualised planning, close monitoring, and ideally a psychiatrist who has thought carefully about timing, taper rate, and what to do if symptoms emerge.
The word deprescribing does not come up often in psychiatry outpatient settings in India. It probably should. A significant proportion of patients on long-term antidepressants were started on treatment for episodes that have since fully resolved. Some are on medications that were never particularly effective but were never reassessed. Others want to stop but have tried once on their own and felt terrible, so they have stayed on the drug, not by choice, but because getting off it seemed impossible.
The IPS multicentric study (Grover et al. 2014), which collected prescription data from 4,480 patients across 11 centres in India, found that approximately 70% of patients with affective and anxiety disorders were co-prescribed a benzodiazepine. Escitalopram was the most commonly prescribed antidepressant, prescribed in over 40% of first-episode depression cases. Antidepressant polypharmacy, defined as two concurrent antidepressants, was present in 8% of prescriptions. These numbers suggest that in routine Indian psychiatric practice, the question of what to stop and when is not hypothetical. It is a daily clinical reality that most clinical encounters do not formally address.
This post explains what deprescribing is, why stopping antidepressants is medically complicated, and how it is actually done based on current evidence.
What Is Deprescribing?
Deprescribing is the planned, supervised process of reducing or stopping a medication that is no longer needed, is causing harm, or where the risk-benefit balance has shifted against continued use. It is defined in the Maudsley Deprescribing Guidelines (2024) as pharmacological regimen optimisation through reduction or cessation of medications for which benefits no longer outweigh risks. The key word is planned. Deprescribing is not the same as stopping medication abruptly, ignoring a prescription, or advising a patient to 'just try coming off it.'
In 2023, the World Health Organisation and the United Nations stated that patients have a human right to be informed of their option to discontinue treatment and to receive clinical support in doing so. Most formal guidance in psychiatry covers how to start medications, with minimal attention to how to stop them. This gap is what the Maudsley Deprescribing Guidelines and a growing body of research literature is now attempting to address.
In clinical terms, deprescribing may mean:
Stopping the medication entirely after a structured taper
Reducing the dose to a lower maintenance level
Switching to a better-tolerated agent before tapering that one
Moving from continuous daily dosing to intermittent or as-needed use
When Should Antidepressant Deprescribing Be Considered?
Not every patient on an antidepressant should come off it. For recurrent major depression, long-term maintenance is often clinically appropriate. The question is not whether to deprescribe everyone, but whether to routinely assess whether each prescription still earns its place.
Deprescribing should be formally considered when:
The original condition is in remission after an adequate maintenance period
The antidepressant never achieved satisfactory therapeutic effect
Intolerable side effects have emerged, such as sexual dysfunction, emotional blunting, or significant weight gain
New drug-drug interactions have arisen that increase risk
The original indication no longer exists, for example, a single mild episode that resolved fully
The patient was prescribed the antidepressant for an indication with limited long-term evidence, such as mild depression or chronic insomnia
The patient has multiple medications and the overall burden of polypharmacy now outweighs the benefit of each individual agent
The timing decision is also important. Deprescribing should generally not be attempted during an acute illness phase, an active substance use period, or a time of major life instability. A patient who has just experienced a significant bereavement, a job loss, or a relationship breakdown is not a good candidate for a taper attempt, however clinically appropriate the deprescribing goal might otherwise be.
Why Stopping Antidepressants Is Not Simple
The discontinuation syndrome
Antidepressant discontinuation syndrome (ADS) refers to a cluster of symptoms that emerge when antidepressants are stopped or tapered too quickly. A 2024 systematic review published in Lancet Psychiatry found that discontinuation symptoms affect a substantial proportion of patients, with venlafaxine and imipramine among the agents with the highest incidence rates. The CANMAT 2024 guidelines state that up to 50% of patients stopping long-term antidepressants abruptly will experience discontinuation symptoms.
Common symptoms include:
Dizziness and imbalance
Nausea and flu-like symptoms
Brain zaps: electric shock sensations in the head or body, often triggered by eye movement
Insomnia and vivid or disturbing dreams
Irritability and mood lability
Sensory disturbances and paraesthesia
For SNRIs specifically, the FINISH mnemonic: Flu-like symptoms, Insomnia, Nausea, Imbalance, Sensory disturbances, Hyperarousal
These symptoms are not signs of addiction. Addiction involves compulsive drug-seeking behaviour and psychological craving. Antidepressant discontinuation is physical dependence: the brain has adapted to the drug's presence and needs time to readjust when it is withdrawn. The distinction matters both clinically and in how we talk to patients about what they are experiencing.
Why venlafaxine is particularly difficult
Venlafaxine (Effexor, Venlor) has a short half-life and consistently shows among the highest rates of discontinuation symptoms in the literature. The Lancet Psychiatry 2024 incidence meta-analysis identifies venlafaxine alongside imipramine as the agents of greatest concern. In clinical practice, patients on venlafaxine often experience symptoms within 24 hours of a missed dose. Paroxetine, with its short half-life and anticholinergic properties, is similarly difficult.
This does not mean venlafaxine cannot be stopped. It means the taper needs to be much slower than many patients and clinicians expect, often taking months, and frequently requiring very small dose reductions in the final stages.
Distinguishing withdrawal from relapse
One of the most clinically difficult aspects of antidepressant tapering is telling apart two things that can look similar: discontinuation syndrome and genuine relapse of the underlying illness. Getting this right determines what to do next.
Discontinuation symptoms typically appear within days of dose reduction, often include physical symptoms like brain zaps and nausea that are not typical of depression, and resolve quickly when the dose is briefly reinstated
Relapse symptoms are more gradual in onset, match the patient's original illness pattern, and do not fully resolve with brief dose reinstatement
The DESS (Discontinuation-Emergent Signs and Symptoms) checklist is a structured tool for assessing discontinuation symptom burden and can help make this distinction more systematic
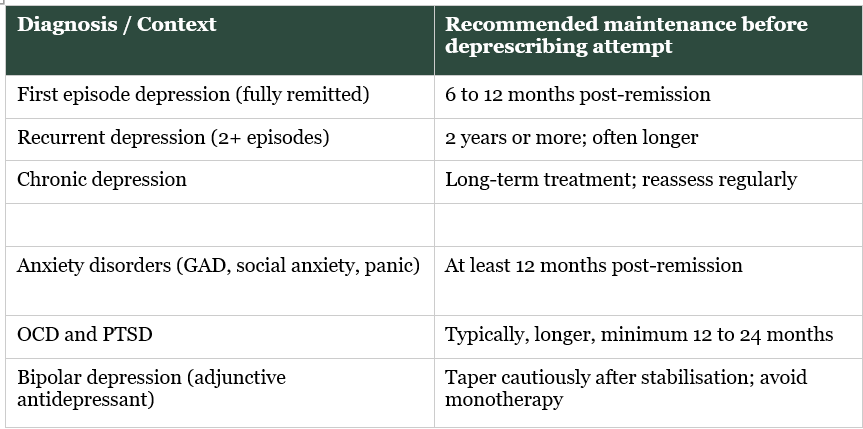
How Long Should You Stay on an Antidepressant Before Considering a Taper?
The recommended maintenance duration before attempting antidepressant deprescribing
These are guidelines, not rules. A patient with three prior episodes, significant psychosocial stressors, and limited access to psychological support should be assessed differently from a patient with one fully remitted episode who has a stable life structure and is engaged in therapy.
How Is Deprescribing Actually Done? The Evidence
The Lancet Psychiatry 2025 meta-analysis
The most clinically important finding in recent deprescribing research comes from a 2025 systematic review and network meta-analysis published in Lancet Psychiatry, which compared deprescribing strategies in adults with remitted depression and anxiety. The key finding: slow tapering combined with psychological support was as effective as continuing the antidepressant at full dose in preventing relapse over the following year, based on moderate-certainty evidence. Abrupt or rapid tapering (under 4 weeks) was clearly associated with higher relapse risk. This is a clinically important finding because it tells us that for patients who are well and want to stop, doing it properly is not a compromise. It is a genuinely viable strategy.
Principle 1: Always taper, never stop abruptly
The evidence against abrupt discontinuation is unequivocal. Even tapers shorter than 4 weeks are associated with worse outcomes than slower approaches. The NICE guidelines recommend slow, stepwise dose reduction proportionate to the current dose. Every guideline converges on this.
Principle 2: Taper slowly, then taper more slowly
This is the most practically important insight from the recent literature. Standard taper advice, such as reducing by 25% every 2 to 4 weeks, is often inadequate for long-term users. Horowitz and Taylor's 2019 paper in Lancet Psychiatry established that serotonin transporter occupancy follows a hyperbolic dose-response curve. This means that cutting from 20 mg to 10 mg of an SSRI has a much smaller effect on receptor occupancy than cutting from 5 mg to 0. The final reductions are pharmacologically the largest and clinically the hardest. A hyperbolic taper, which slows the rate of reduction as doses get lower, minimises the biological disruption at each step and is now recommended for patients with difficulty stopping. The principle: stop slow as you go low.
In practice, this often requires liquid formulations or specially compounded doses to achieve dose reductions of 1 to 2 mg at the lower end of the taper. This cannot be done reliably by cutting tablets. Coordination with a pharmacist is necessary, and supervision by a clinician familiar with deprescribing is essential.
Principle 3: Pair tapering with psychological support
The 2025 Lancet Psychiatry meta-analysis found that psychological support, whether CBT-based relapse prevention or structured counselling, significantly improved outcomes during tapering. This is not optional for higher-risk patients. Building psychological resilience during the taper period, rather than after, is part of the deprescribing plan.
Principle 4: Personalise the plan
The rate of taper, the choice of liquid versus tablet formulations, the frequency of review appointments, and the decision to add psychological support all depend on the individual. Relevant factors include:
Duration of antidepressant use (longer use typically requires slower taper)
The specific drug (venlafaxine and paroxetine require particular care)
Previous taper attempts and how they went
Current life circumstances and available social support
Original diagnosis and relapse risk profile
Principle 5: When to manage discontinuation symptoms
If symptoms emerge during a taper, the first step is to slow or pause the reduction. If symptoms are severe, reinstating the previous dose briefly and attempting a slower taper is standard practice. In some cases, switching to fluoxetine (which has a long half-life and lower discontinuation risk) before tapering can be a useful strategy. Symptomatic management may include:
Paracetamol or ibuprofen for headache
Ondansetron for nausea
Short-term benzodiazepines for significant anxiety or insomnia, used cautiously and for a defined period
Liquid formulations for finer dose adjustments in the final taper stages
The Long-Term Antidepressant Problem
The patient who has been on an antidepressant for 10 or 15 years, has had little or no discussion about stopping, tried once on their own and felt terrible, and subsequently stayed on the drug not by choice but by apparent necessity: this is more common in practice than clinical literature tends to acknowledge.
Data from the UK show that nearly half of all antidepressant users take them for more than two years. In India, the IPS multicentric study (Grover et al. 2014) does not capture long-term follow-up, but the high rates of co-prescribed benzodiazepines, which often begin as short-term anxiolytics and become chronic, suggest that therapeutic inertia around psychotropic continuation is a real phenomenon in Indian practice too.
For long-term users, hyperbolic tapering with liquid formulations is frequently necessary. The process takes months to years in some cases. That is not a failure of the patient or the clinician. It is a consequence of the brain's adaptation to a drug taken continuously for a long time, and a reason why the process needs to be planned, monitored, and paced.
There are also real long-term risks of antidepressant use that are worth acknowledging honestly, including sexual dysfunction (estimated to affect 30 to 40% of SSRI users), emotional blunting, weight gain particularly with paroxetine and mirtazapine, and in some patients, a worsening of mood over time despite continued treatment. These are clinically legitimate reasons to revisit the continuation decision, not arguments against antidepressants as a class.
A Note on My Approach to Deprescribing
In my clinical practice, deprescribing is not something I view as abandoning treatment. I see it as completing it. When a patient has recovered from depression, maintained stability for an appropriate period, built psychological resilience, and wants to try life without the medication, supporting that process carefully is part of responsible psychiatric care.
It also matters that we are honest about what antidepressants cannot do. They are not indefinitely risk-free. They are not always easy to stop. And for patients who have been on them for years without a formal review, the question of whether they still need them deserves a careful answer.
If you or someone you care about is thinking about stopping an antidepressant, do not attempt it without clinical support. The process is manageable, but it needs to be planned individually. You can book a teleconsultation through ManoMitra to discuss whether deprescribing is appropriate for your situation.
Frequently Asked Questions
How long does antidepressant deprescribing take?
It varies considerably. For patients who have taken antidepressants for less than a year without previous difficulties, a taper over 4 to 8 weeks may be sufficient. For those on long-term, high-dose venlafaxine or paroxetine, the taper may take 6 to 12 months or longer. There is no fixed schedule that works for everyone.
Is stopping an antidepressant the same as addiction withdrawal?
No. Antidepressant discontinuation syndrome is physical dependence, not addiction. Addiction involves compulsive drug-seeking, craving, and continued use despite harm. Antidepressants do not activate dopamine reward circuits in the same way. The discontinuation symptoms are real and can be severe, but they reflect neuroadaptation, not addiction.
Why did my previous attempt to stop go badly?
A previous failed attempt almost always means the taper was too fast. The brain had adapted to the presence of the drug over months or years, and the reduction rate did not allow enough time for readaptation. A much slower, more carefully structured approach changes the outcome for most patients. A prior bad experience is information, not a sentence.
Can I cut my tablets in half to taper?
Tablet cutting gives rough dose reductions and is not reliable for the precise reductions needed at the lower end of a taper. For standard reductions in the early stages, it may be acceptable. For the critical lower-dose stages of a hyperbolic taper, liquid formulations or compounded doses are usually needed. This requires a prescription and pharmacist involvement.
Will I relapse if I stop my antidepressant?
It depends on your history. For a fully remitted first episode, the risk is substantially lower than for recurrent depression. The 2025 Lancet Psychiatry meta-analysis found that slow tapering with psychological support was comparable to continuing the antidepressant in preventing relapse for people who were fully remitted. This is an individual discussion that belongs in a clinical consultation, not a general answer.
Is deprescribing relevant in India?
Yes. The IPS multicentric study (Grover et al. 2014) found that benzodiazepines were co-prescribed in approximately 70% of patients with affective and anxiety disorders, and antidepressant polypharmacy was present in 8% of prescriptions. These prescribing patterns make systematic medication review and deprescribing a clinical competency that is needed in routine Indian psychiatric practice, not only in specialist settings.
Explore More on This Topic
Antidepressants and Sexual Side Effects: What Patients Should Know
Serotonergic Psychedelics as Antidepressants: What the Evidence Shows
Conditions We Treat at ManoMitra
Dr. Anindo Mitra is a Consultant Psychiatrist at Athena Behavioural Health, Gurugram. He completed his MD in Psychiatry from JIPMER, Puducherry. His clinical focus includes evidence-based pharmacotherapy, deprescribing, and the neurobiology of psychiatric disorders. He writes at dranindomitra.com on mental health education for the Indian public.
This post is for educational purposes only and does not constitute individualised medical advice. If you have concerns about stopping or adjusting your medication, please consult a qualified clinician.